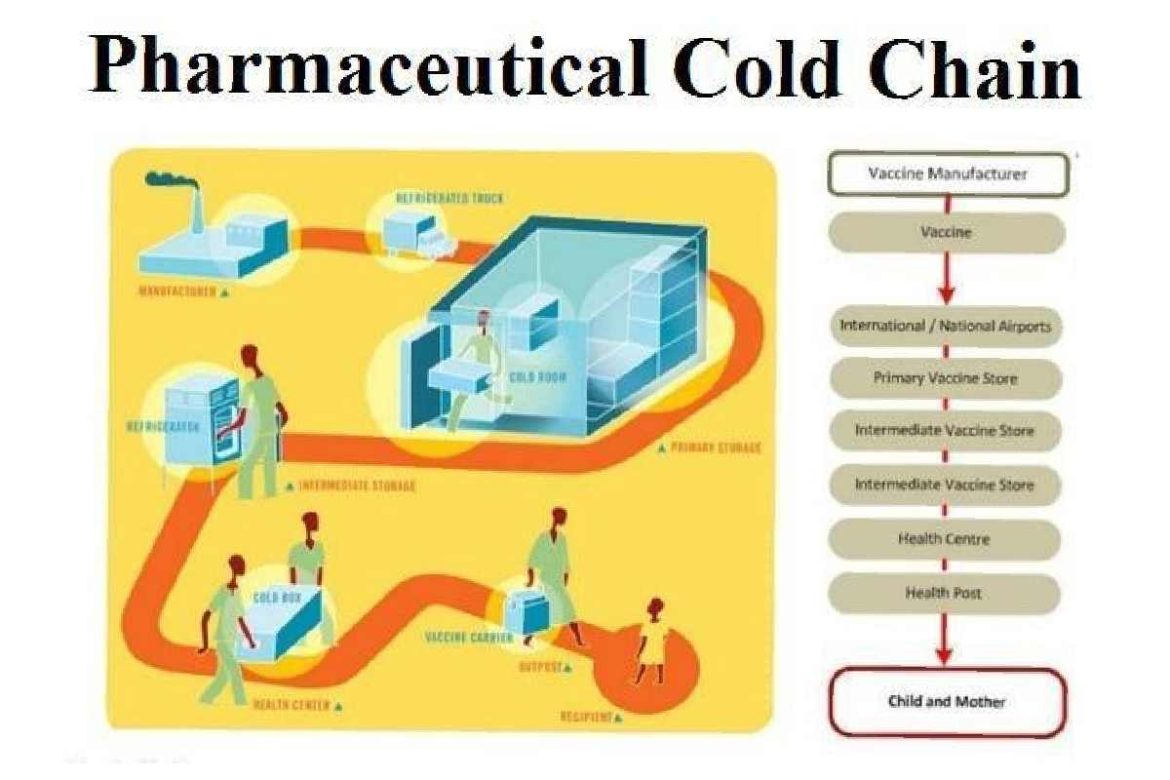
The Pharma Industry Booms Thanks to The Cold Supply Chain – The cold chain is a vital part of the pharma industry. As the need to maintain medications in carefully monitored, low-temperature environments increases as the US Food and Drug Administration (FDA) approves more such meds, the cold chain market is experiencing rapid growth. In this article, we will look at the current market conditions, explore what is driving the growth and where competition exists, and look at the regional leaders in that market.
Table of Contents
The Current Market Conditions
Advances in pharmaceutical consulting, research, development, and products have increased the need for cold chain logistics and proper storage of temperature-sensitive assets. Many new vaccines and medications require low-temperature environments during transportation and storage to remain effective. The fragility of these assets has changed how they are shipped. Though this has created challenges in cold chain technology, two main factors have improved the success of moving products through the cold chain and maintaining their integrity.
First, proper freezer units have helped ensure product integrity. Manufacturers now construct and distribute low-temperature units designed to ensure safe storage of sensitive assets. Another tool that has contributed to maintaining cold chain integrity is the data logger. When cold chain stakeholders use both low-temperature freezer units and data loggers, it is far easier to meet compliance guidelines and respond to issues as they may occur.
Why The Cold Chain Is Essential
The pharmaceutical and healthcare sectors require a safe and unbroken cold chain because products produced by this industry are vital. And their safety and efficacy rely on temperature. When temperature-sensitive medications warm to unsafe levels, they may be rendered useless. This results in not just inventory loss, but financial loss as well. Big pharma is big business and downtime is costly. This puts additional pressure on the need for a functioning and reliable cold chain. Without it, medications cannot get where they are needed. And that broken cold chain may contribute to spreading disease rather than controlling it.
The Driving Forces
The development of new medications with greater efficacy is the result of growth within the pharmaceutical and healthcare sectors. These new types of drugs require storage and refrigeration solutions that were not common just a few years ago, and their requirements for temperature-controlled environments are driving the new cold chain. In addition, there has been a rapid increase in pharmaceuticals export worldwide. Some countries lack drug manufacturing facilities. They may also lack the means to maintain temperature compliance for sensitive medications. These factors are driving the future of the cold chain into parts of the world where it previously didn’t exist.
Vaccines developed and distributed to control the spread of COVID-19 are an example of how a worldwide cold chain has developed. Governing bodies have worked together for mass immunization of highly vulnerable populations. Doing so requires the storage of massive quantities of vaccines that require temperature-controlled environments to remain effective until they are administered. The pandemic disrupted the supply chain but also brought to light the need for better solutions for transporting and storing such sensitive assets as pharmaceutical products. All these forces in play have contributed to driving development in maintaining a solid cold supply chain.
The Competition
The key players responsible for the logistics of the cold chain are typically companies known for their ability to transport goods quickly and efficiently. But the increase in the volume of sensitive pharmaceutical assets that need transporting shows a need for more players in the transportation and storage industry. Safe low-temperature environmental conditions for many of these new medications will produce new, innovative competitors in the market, with storage products designed to address the environmental conditions sensitive assets require.
The Regional View
The lead in the healthcare cold chain logistics market would go to North America at this point, because some of the largest pharmaceutical companies and suppliers are based in North America. Ongoing growth within that market will continue to solidify the market share. However, Europe and the Asian Pacific regions may become contenders for the top spot based on factors such as market participants, government programs, and a well-designed and functioning healthcare system.
The Future
The cold supply chain will continue to grow, given the ongoing development of medications that require storage in low-temperature settings. Dickson believes that products that address these assets’ environmental needs will improve and cold storage for pharmaceuticals solutions will expand to include new players vying for part of the action. With few players currently in the marketplace, additional competition may assist in the development of newer, more efficient products that can address as yet unidentified or unrecognized cold chain needs. This is an exciting market to be part of and will see growth well into the future.
Final Thoughts
Cold chain technology has come a long way. Ultra-low-temperature storage and transport containers are now designed specifically for cold chain use. Monitoring tools, like data loggers, help ensure compliance with temperature requirements. Technology has helped stabilize cold chain transport and make massive improvements as the cold chain becomes a vital part of the pharmaceutical and healthcare sectors.